 
If ∆S is negative, then the negative signs (from the subtraction and the sign of ∆S) will cancel out, and so as T∆S gets bigger, ∆G will get more positive. T is always positive, so if ∆S is positive then a bigger T∆S will make ∆G more negative (since we subtract T∆S). As T increases, the T∆S component gets bigger. ∆H is still positive and ∆S is still whatever sign you figured out above. Since ∆H and ∆S don't change significantly with temperature (given in the question), we can assume that they keep the same signs and values: i.e. If ∆G is negative (from the question), is the reaction spontaneous or non-spontaneous?Ģ) Let's use ∆G = ∆H - T∆S again. From these values, we can know for certain whether ∆S is positive or negative (hint: remember that we are subtracting ∆G!).ġ) Knowing the sign of ∆G is enough to say whether the reaction is spontaneous or not under these conditions. Temperature is always positive (in Kelvin). We know (from the question) that ∆G is negative and that ∆H is positive. (4) CO 2(g) -> CO 2(s) Aug 2004- 21 Even though the process is endothermic, snow can sublime.This looks like a homework question, so I'll give you some hints to get you on the riht path rather than answering directly.ģ) We know that ∆G = ∆H - T∆S. Jan 2005- 43 Which of these changes produces the greatest 
June 2005- 20 Systems in nature tend to undergo changes toward (4) decreases because the particles are more randomly arrangedĪug 2005- 46 At STP, a sample of which element has the highest entropy? (3) decreases because the particles are less randomly arranged (2) increases because the particles are more randomly arranged (1) increases because the particles are less randomly arranged Jan 2007-27 In terms of energy and entropy, systems in nature tend to undergo changes towardĪug 2006- 49 Which list of the phases of H 2O is arranged in order of increasing entropy?Īs a sample of I 2(s) sublimes to I 2(g), the entropy of the sample (4) The dissolving of the LiBr(s) in water is an exothermic process. (3) The dissolving of the LiBr(s) in water is an endothermic process. (2) The entropy of the LiBr(aq) is less than the entropy of the water. (1) The entropy of the LiBr(aq) is greater than the entropy of the water. View Available Hint (s) Reset Help 1 mol of 1 mol of carbon disulfide gas at 273 K and 40 L 1/2 mol of 1/2 mol of 1/2 mol of 1 mol of 1 mol of liquid xenon at 100 K xenon gas at 273 K and. To rank items as equivalent, overlap them. Which statement best explains why the thermometer reading initially increases when LiBr(s) is dissolved in the water? Transcribed Image Text: Rank these systems in order of decreasing entropy. 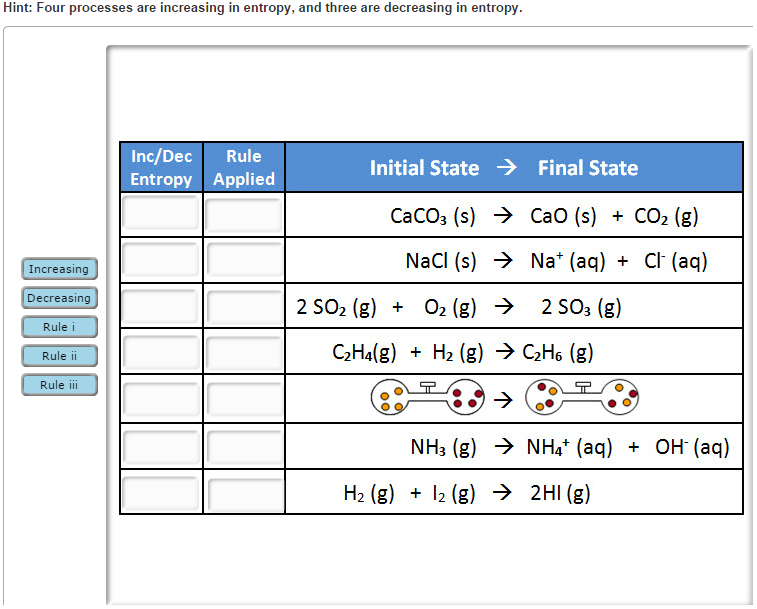
June 2008- 17 A thermometer is in a beaker of water. June 2008- 45 Which 1-mole sample has the least entropy? (4) It is exothermic, and entropy increases.Īug 2009- 45 The entropy of a sample of H 2O increases as the sample changes from a (3) It is exothermic, and entropy decreases. (2) It is endothermic, and entropy increases. (1) It is endothermic, and entropy decreases. Jan 2010 44 Given the balanced equation representing a phase change: SeptemChristian Right Lobbies To Overturn Second Law Of Thermodynamics Solid and liquid becomes a solution and gasĬ 2H 5OH(l) + 3O 2(g) -> 2CO 2(g) + 3H 2O(g) A process where a solid changes to a liquid or gas or a liquid changes to a gasĮxamples-Highlight the boxes to reveal the answer and reasoning.Ģ NaHCO 3(s) -> Na 2CO 3(s) + H 2O (g) + CO 2(g)ĢNH 3(g) + CO 2(g) -> NH 2CONH 2(aq) + H 2O(l)ĢNa 2O 2(s) + 2H 2O(l) -> 4NaOH(aq) + O 2(g).A reaction occurs in which there is an increase in the moles of gas in the product.A reaction breaks up a larger molecule into smaller molecular fragments.***Entropy increases will be observed when any one or more of the following occur: The entropy change of a reaction is given the symbol ΔS, units when used are J/mol K (not kJ). For instance a mixture of two different gases will have greater entropy than the sum of the entropies of the two gases alone. In this example there are two moles of gas on the right hand side and only one mole of gas on the left hand side - the entropy increases going from left to right (reactants to products).ĭisorder can also arise by mixing different components. If the number of moles of gas increases in a chemical reaction (as shown by the stoichiometry of the reaction) then the entropy also increases. Solids < Liquids < Aqueous Solutions < Gases Solutions have a greater entropy than pure liquids as the particles in a solution are more separated and solvent molecules separate the solute particles. Liquids have much lower entropies and solids lower still. Gas particles have random motion have high entropy values. 
This is considered to be the degree of disorder of a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
